 The electron shells between the outer electrons and the nucleus have a ‘shielding’ effect which reduces the attraction of the outer electrons to the nucleus. The number of shells of electrons also increases. And nuclear charge increases hands ionization and no Taji increases. Reactivity: Very Reactive most reactive group on the Periodic Table. How does the reactivity of Group 1 metals change As you go down a group the atomic number increases. So hands tendency two lose electron increases in group now across the period that is from left to right. The more the nuclear charge, the higher the nuclear charge the higher will be the ionization energy and the lower the nuclear charge the lower will be the ionization energy. Now ionization energy is directly proportional to the nuclear charge. Both are correct decreases vital because ionization energy is the energy required to lose the electron from the outer most shell. So nuclear charge decreases and ionization and tell P or we also call it as ionization energy. So now we know that down the group number of shells increases hands atomic radius which we often term as atomic size also increases. In general, atomic radius decreases across a period and increases. The more the tendency to gain electron, the more nonmetallic the element is. Atomic radius is the distance from the atoms nucleus to the outer edge of the electron cloud. The more the tendency to lose the electrons, the more metallic the element is nonmetallic tendency is the tendency to gain electron. As we go across a period from left to right, we add a proton to the nucleus and an electron to the valence shell with each successive element. What do we mean by metallic or nonmetallic character? Metallic character is the tendency to lose or release electrons. First ionisation energy increases across the period. Period 3 shows trends in atomic properties: Ionic radius decreases across the period. It contains the elements sodium, magnesium, aluminium, silicon, phosphorus, sulphur, chlorine, and argon. They have comparable characteristics because the number of valence shell electrons is the same.First of all. Period 3 is the third row in the periodic table. The electrical configuration of the valence shell of the elements is repeated in such a manner that elements with the same valence shell electrons fall under the same group when they are arranged in order of increasing atomic number. 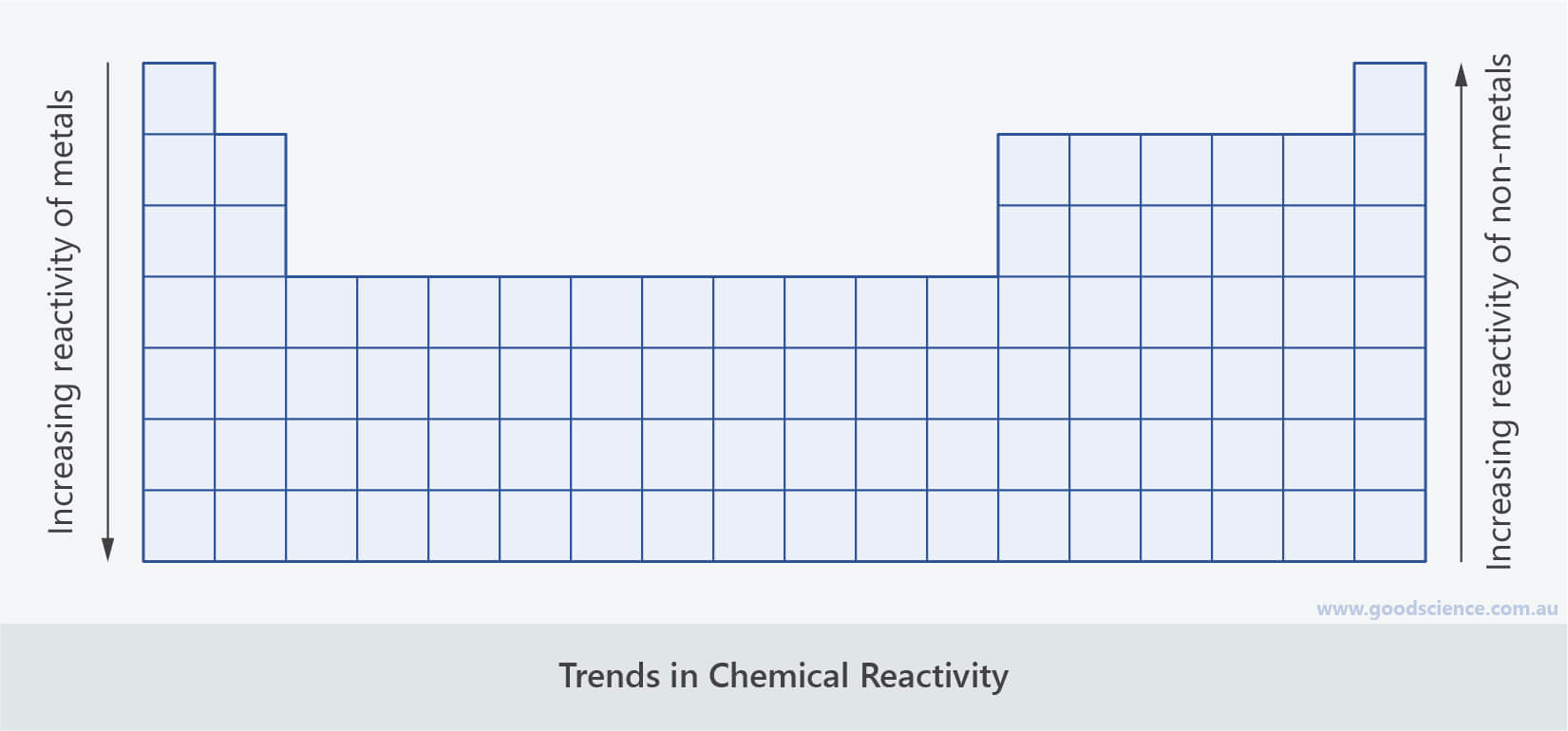
The electrical configuration of the elements provides the basis for the current periodic table. The higher the electronegativity, the farther right and up on the periodic table you go. The stronger the electronegativity, the more intense the electron exchange. Over the course of a time, reactivity rises as you move from left to right.Īs you move down the group, your reactivity declines. The simpler it is for electrons to be given or taken away the farther to the left and down the periodic chart you travel, the greater the reactivity. Period - As you go from left to right throughout a period, your responsiveness diminishes.Īs you go down a group, your reactivity rises. Because the transfer/interaction of electrons is the foundation of chemical processes, this is generally defined by how readily electrons can be withdrawn (ionisation energy) and how strongly they desire to take other atom's electrons (electronegativity). In this way the Periodic Table can be used to predict how a particular. The reactivity of an atom relates to how probable it is for it to react with other substances. Reactivity decreases going down Group VII Melting point decreases going down Group I. That is why elements in the periodic table have periodic physical and chemical characteristics. 
Elements are placed in the contemporary periodic table according to their atomic numbers, which, as previously established, are closely connected to their physical and chemical characteristics. Order of Periodic Trends and Reasons : When we go left to right atomic radius and ionic radius decreases.  
Hint: “Physical and chemical characteristics of the elements are periodic functions of their atomic numbers,” according to Moseley's Modern Periodic Law. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
